Retrospection - by Prof. Tom Reimchen (University of Victoria, BC, Canada)
Ric’s research exposed me to methods of studying variation in natural populations. One of the techniques involved gill-netting cutthroat trout from the lake, extracting the stickleback from the stomachs and then measuring their size and various defense traits including the bony lateral plates on the side of the fish. Ric got the impression that stickleback with seven lateral plates, which comprised the modal (most common) phenotype in the population, were captured by trout at a lower rate than stickleback with fewer or greater number of plates. He suspected that fish with different numbers of plates differed in their behaviour and that this variation might account for the trends in trout stomach contents. He assigned me the task of locating stickleback nests in the shallows of this large lake, after which we would capture the territorial males. Ric scored the males for nuptial colour, number of lateral plates on both sides, and body size. I did not really understand the rationale for this but it gave me the first exposure to the idea that individual phenotypes differing by a single lateral plate might be acting in different ways.
Over the three summers that I worked as Ric’s assistant, I found many nests and Ric scored many male stickleback. Ric thought it might be interesting to sample some of the neighbouring lakes to see if there were any other interesting stickleback. We hiked into the interior of the island across Sphagnum bogs and through forests using survey maps and compass bearings occasionally missing lakes altogether. One of these isolated lakes was much smaller than Mayer Lake, lacked littoral vegetation, and was appropriately named Drizzle Lake. As we walked along the shoreline, speculating on whether there would be any fish in this small lake, I found a dead stickleback on the shore that looked superficially like the giant black Mayer Lake stickleback. I thought Ric had dropped a Mayer Lake stickleback to fool me into thinking that we had discovered another example of gigantism. He assured me that he had done no such thing. The genetic work years later showed that this was an independent origin of gigantism from that at Mayer Lake. On inspection of this dead stickleback, we could see that it had only 4 lateral plates, lower than the lowest plate count that Ric had ever seen at Mayer Lake, and we concluded (naively) that there would be minimal predation in the lake.
In my free time, I hiked to some additional unsampled lakes. One of these was the small Boulton Lake that lacked predatory fish. I put some traps in, waited a few hours, and when I checked them, to my absolute amazement, I found that most of the stickleback had no pelvis and many were missing some of the dorsal spines. Excitedly I returned to Mayer Lake to tell Ric who told me that such a stickleback had not been previously found in North America or Europe. We continued these lake surveys and got stickleback from 22 lakes, and many of the populations were distinctive to each lake. Such variability across such short distances exceeded the known morphological diversity of stickleback throughout Canada and Europe. The provincial environmental agency got wind of our ‘discoveries’ and asked us to recommend three lakes with unusual stickleback, one of which would be established as an Ecological Reserve for long term protection of the entire watershed and opportunity for research. We proposed Mayer Lake, where Ric was doing his study, Drizzle Lake, with the other giant stickleback in the north of the island, and Boulton Lake, which had stickleback with a missing pelvis. The government made a decision that Drizzle Lake would be established as an Ecological Reserve as it had the fewest administrative conflicts with other agencies. I was happy with this decision as the undisturbed lake was remote with no road access and had a small old log cabin near the lake that would provide a living place if I ever were to do research on the fish in this lake.
In May 1970, after completing my Zoology undergraduate degree at the University of Alberta, I convinced my friend Joe Rasmussen to come with me to Haida Gwaii for several weeks to sample more lakes for stickleback. During this trip, we discovered a small acidic bog pond with stickleback that were very unusual: not only were the lateral plates missing but the entire trunk was covered in a unique dinoflagellate parasite. We called this locality Rouge Lake as the nuptial colour of the males was outlandishly red; it turned out with later studies to be also very atypical with respect to its genetic structure. On this expedition, I also made detailed collections of the Boulton Lake stickleback and saw that the relative expression of the pelvic girdle differed from one part of this lake to another, variation that I was later able to relate to spatial differences in the predation landscape.
During the spring of 1970, I started looking at samples of the giant stickleback from Drizzle Lake and noticed that some of the anterior lateral plates underlay the basal support structures for the dorsal and pelvic spines. One atypical fish had spines that were easily laterally deflected from their erect position. This fish was missing one of the lateral plates and it immediately became clear that the plates would buttress the dorsal and pelvic spines from lateral forces exerted during predator manipulation. I shelved my idea about this for several years, returning to it in the late 1970’s and eventually publishing these observations in 1983 (Evolution 37: 931-946 - Figure 1).
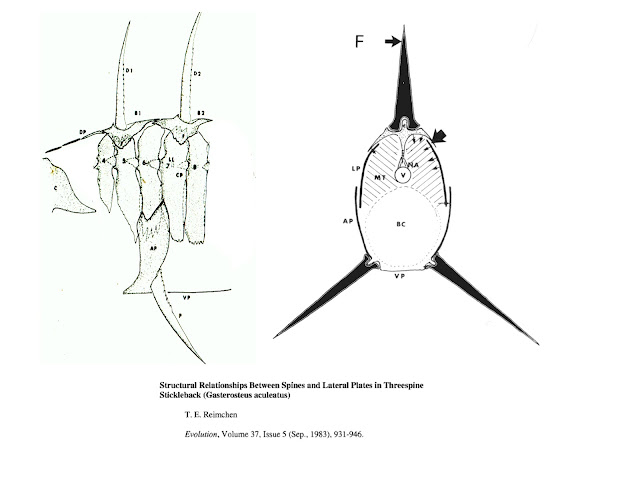 |
| Figure 1: Relationships between lateral plates and spine supports. |
Ric Moodie suggested I read EB Ford’s recent book on ecological genetics. This completely hooked me on studying adaptation and measuring selection in real time in the field. Fortunately, the University of Alberta had just hired Kennedy McWhirter, who was Ford’s colleague at Oxford. I took the courses Ecological Genetics and Population Genetics from Kennedy and this further cemented my interest in this emerging discipline. Kennedy encouraged me to do graduate work at the University of Liverpool in the UK where Arthur Cain and Philip Shepard had recently arrived from Oxford and were developing a graduate program in Ecological Genetics. My subsequent four years in the UK resulted in a DPhil on the ecology and genetics of two sibling species of intertidal gastropods. This research greatly emphasized to me the importance of spatial and temporal scale for interpreting polymorphic variation within and among populations, an approach that would structure my subsequent studies on stickleback.
During my graduate program, I became friends with Paul Handford and Graham Bell who were both at Oxford; over several years, we hatched an outlandish research plan in which we would go to Drizzle Lake on Haida Gwaii, and with our partners, live in an old log cabin, and undertake a multi-year study on the giant stickleback from the lake; Paul would focus on the behavioural adaptations of the fish, Graham on the demography of the population and myself on predator-prey interactions in relation to phenotypic variability. Paul joined me on Haida Gwaii in late 1975 and 1976. Paul, who had just finished a post-doc on songbird dialect in the dry mountains of Argentina, did not take well to living in a small dark cabin in the midst of a wet and cool rainforest. Graham at this time had just finished his DPhil at Oxford and with Sue, his partner, initiated the process of immigrating to Canada. They got as far west as Edmonton where Paul was staying. It was clear that the logistics of potentially six of us living at Drizzle Lake were unreasonable. Paul and Graham got sensible and each got jobs, Paul at the University of Western Ontario and Graham at McGill University.
 |
| Figure 2a. Haida Gwaii with Drizzle Lake and research cabin (inset) |
Not as well-anchored in reality, I took up residency at the Drizzle Lake cabin in 1976 (Figure 2a) and began the research program. I wanted to extend principles from some of my thesis work involving polymorphic traits and felt that sources of mortality and microsite adaptations might also be operating with traits that were continuous, such as lateral plates, rather than only discontinuous traits like the colour of intertidal snails. With a $2000 support from the Ecological Reserves Unit (BC government) as well as similar amounts from Joseph Nelson at the University of Alberta, my partner Sheila Douglas and I equipped the 80 year old Drizzle Lake cabin with solar panels, a small wind generator, lights, microscopes as well as a ‘portable’ Osborne computer. In 1985, I was successful at getting an NSERC operating grant that allowed us to continue the research.
 |
| Figure 2b. The lab at Drizzle Lake. |
Our year round residency at Drizzle Lake from 1976 to 1985 and then summer residency from 1986 to 1990 yielded substantial evidence of repeatable temporal and spatial variation in the predation regime and the potential influence on the selection landscape affecting the stickleback population. Within several years, we had identified over 20 species of predators on stickleback in the lake, including 16 species of avian piscivores as well as resident salmonids (Figure 3). Most of these predators differed seasonally and they differed as to where they foraged in the lake and what size classes of stickleback they consumed. This high diversity of predators was not because the lake was a predator ‘hotspot’ but rather, evidence for this diversity emerged only as a consequence of the extended time duration (multiple years, multiple seasons) of the study. I summarized these data in a chapter for an Oxford publication that Mike Bell and Susan Foster put together in 1994.
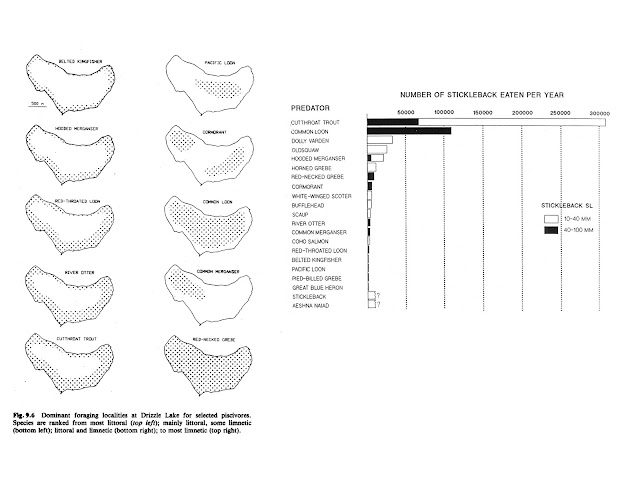 |
| Figure 3: Predator assemblage at Drizzle Lake. |
During the research program, Sheila and I circumnavigated Haida Gwaii several times and sampled about 800 lakes, ponds and streams, of which about 15% had stickleback, all of which were morphologically different from each other. Each lake offered a distinctive set of biophysical parameters (predators, diet, parasites, lake morphometry, spectra, etc) that had the potential of structuring the selective landscape (Figure 4).
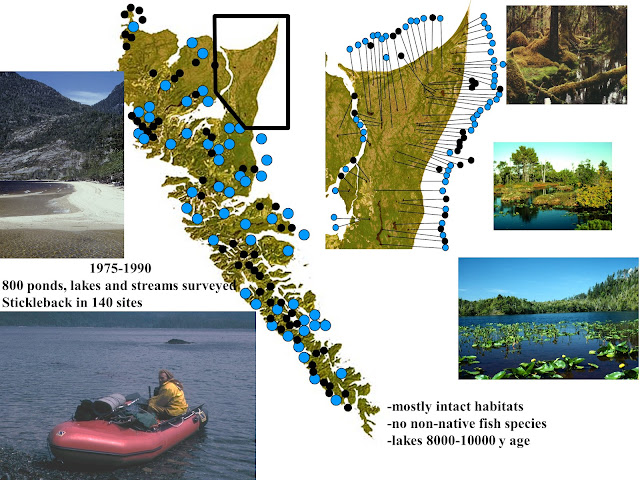 |
| Figure 4: Geographical survey of Haida Gwaii for stickleback with representative examples of habitats. Symbols: blue- stickleback present, black- stickleback absent |
I was able to recruit some excellent students including Carolyn Bergstrom on the role of asymmetry in defensive traits, Patrik Nosil on fluctuating selection and Mark Spoljaric on plasticity and predictability of body shape. We summarized the adaptive radiation of these 100+ allopatric populations in 2013 (Evolutionary Ecology Research 15: 241–269). Essentially, the selective landscape at each locality was defined by the relative importance of puncturing, compression or grappling piscivores combined with water spectra (reaction distance) and lake size (Figure 5).
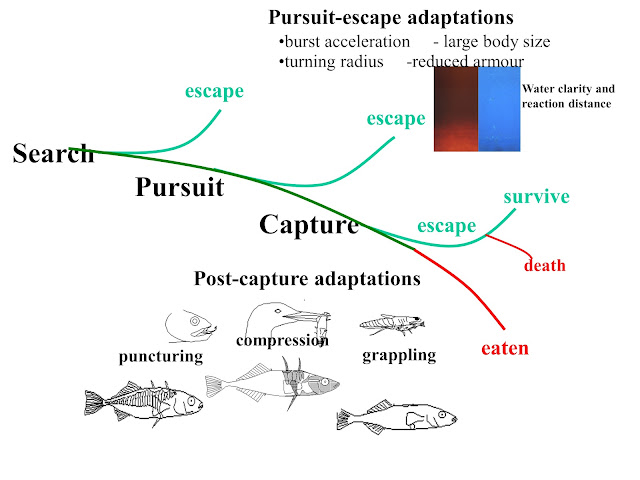 |
| Figure 5: Summary of major processes influencing the selective landscapes of Haida Gwaii stickleback defense structures. |
While it was clear to me that large differences in morphology, from fully-armoured to unarmoured among and within these populations, were adaptive; it remained ambiguous whether this represented genetic variation or plasticity. Axel Meyer had recently shown a major role of adaptive plasticity in jaw and skull morphology of central American cichlids, and such plasticity could not be ruled out for these divergent Haida Gwaii populations. In 1989, I asked a new graduate student, Patrick O’Reilly, to examine the mtDNA of some of the most divergent populations of stickleback, including the unarmoured stickleback in small acidic ponds that I discovered back in 1970. It was reassuring that these unarmoured fish had a very distinctive mtDNA haplotype from most other stickleback populations (although it was similar to those in Japan), and were potentially relictual but this did not resolve whether the unarmoured phenotype was heritable. Subsequently, advances in DNA sequencing techniques allowed David Kingsley at Stanford to develop genome-wide SNP arrays for stickleback. My post-doc, Bruce Deagle, was able to use the arrays to show extensive genomic differentiation among the morphologically divergent populations, including three different lake-stream species pairs (Proc. Roy. Soc. 279 : 1732 1277), although this did still not rule out adaptive plasticity for divergence in morphological traits.
The repeated evidence for adaptive differentiation among and within populations would have more conceptual context if I had some experimental data that allowed estimates of strength of selection and rates of change. In the mid-1980’s, I sketched out a plan to transplant limnetic giant stickleback with robust armour from a large lake into a small ‘barren’ fishless pond that differed in multiple ecological axes from the source lake. Ideally, I wanted to create a shift in the ecological theatre involving predation landscape (salmonids/birds to macroinvertebrates), trophic regime (limnetic/plankton to benthic/macrobenthos), spectral regime (dystrophic/heavily stained to eutrophic/non-stained waters) and water chemistry (lower to higher conductivity), expecting that subsequent generations might reveal phenotypic changes in the direction predicted from the differences I observed in the geographical surveys (Figure 6). Eventually, I identified four suitable barren ponds that generally met the criteria and in 1992 initiated the first of the transplant experiments.
 |
| Figure 6: Transplant experiment from Mayer Lake with giant stickleback into a barren roadside pond. |
The colonists successfully reproduced, yielding numerous generations that I sampled over time. In 2007, an outstanding student (Steven Leaver) began a graduate program with me and photographed and measured all the samples for meristic and metric traits. The results produced striking evidence for shifts in all defense and trophic traits over nine generations, all in the direction predicted. Some of the traits shifted in the first generation, consistent with adaptive plasticity, and other traits shifted across generations, more consistent with genetic changes (Biol. J. Linnean Society. 107:494-509 - Figure 7). David Kingsley offered to do whole-genome sequences and I sent him 56 stickleback, including the source and transplant populations as well as representative stickleback from morphologically divergent Haida Gwaii populations. I needed a post-doc to do the genomics of these samples and fortunately Katie Peichel encouraged David Marques to apply. Remarkably skilled, David was able to complete the analyses of these fish identifying trait-specific genetic markers across the genome. These results gave novel insight into opsin evolution, as well as evidence for genome-wide shifts, all in the direction that were predicted by the genomic differences among the allopatric populations differing in ecological conditions. I feel this paper (Marques et al. 2017, Nature, Ecology and Evolution) has been the most substantive to emerge from my research program as it exemplifies the efficacy of natural selection and predictability of evolutionary changes among populations in remarkably few generations. This theme greatly contrasts to that of the famous orator, Stephen J. Gould, who concluded in 1985 that “….some geographic variation within a species is clearly adaptive, but much is a non-adaptive product of history."
 |
| Figure 7: Results of phenotypic shifts in the transplant population relative to the original colonists after 8 generations. |
One of the important concepts that emerged during our many years in the ecological theatre at Drizzle Lake and Haida Gwaii was the persistent reminder that inter- and intra population variability in defense morphology could not be reliably understood without context to age-specific sources of mortality. Presence of multiple species of piscivores that differed spatially and seasonally, as well as in their foraging and prey capture behaviour, creates an opportunity for diversifying selection on the traits that can differ spatially within populations and fluctuate over short periods of time. The tendency of researchers, including Ric and myself in the early formative years, to classify populations as either with or without predators was simply wrong. There were localities without predatory fish but there were none that lacked one or more species of piscivorous birds or macroinvertebrates (Figure 8).
Researchers, editors and reviewers still uncritically accept the flawed dichotomous characterization of the predation landscape (yes/no or high/low) despite the lack of evidence to warrant the dichotomy. While It is unlikely that future researchers will embrace my approach - living in an old cabin at the study lake for year-round observations on the abundance and behaviour of the predator assemblage, I am optimistic for the future. New technologies, such as hi-resolution field cameras and e-DNA will hopefully contribute to the understanding of the subtle but real temporal and spatial variability in selective landscapes, the motivation for the extended field studies on Haida Gwaii stickleback.
 |
| Sheila Douglas putting in the work. |






No comments:
Post a Comment