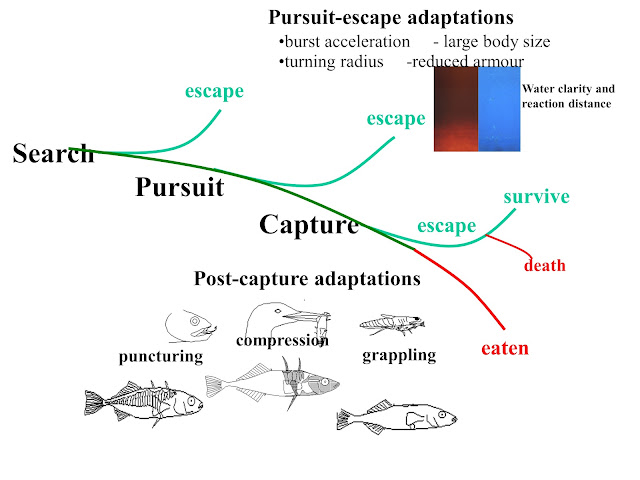
When a colleague and friend passes on, those of us left behind wish to honor them with some of our favorite memories. With Ole Kristian Berg, those memories are many and vivid. He was an excellent colleague and a genuinely wonderful man, who not only brought a creative originality to his (and our) research but also an undimmed sense of wonderment about the natural world, especially salmonid fishes (salmon, trout, and charr). Most importantly, he was someone who loved and lived life to the fullest, and never ceased to help us all remember how lucky we are to have careers and lives that can be so fun.
This post has memories from myself (Andrew Hendry), Sigurd Einum, Thomas Quinn, Trond Amundsen, Gunnbjørn Bremset, Jan
Grimsrud Davidsen, Tor G.
Heggberget, Sten
Karlsson, Line
Elisabeth Sundt-Hansen, and Eva Marita
Ulvan - as well as (at the end) a collection of Ole's most cited papers. Note: many more remembrances from family, friends, and colleagues are HERE in Norwegian.
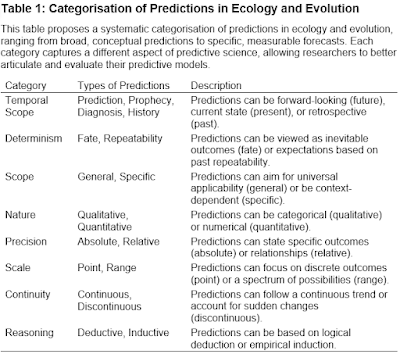
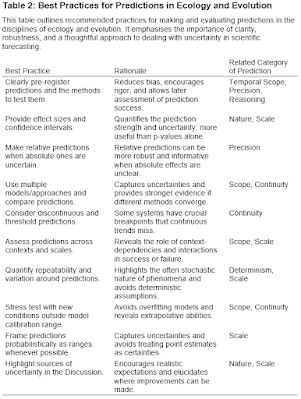
 |
| Ole Kristian Berg (11.04.1954 - 26.02.2023) |
Andrew Hendry
I was a pretty green first-year graduate student in 1993 when I first met Ole - and I remained his close friend and collaborator for another 10 years. I worked with him four different years in Alaska (over the span of 10 years) and I spent months (over several trips) with his family in Trondheim, Norway. He was my first real international collaborator - and a more wonderful and personal entre into such collaborations could not have been imagined. Ole was an important and innovative scientist, with some truly influential work that you can peruse below. Ole was about so much more than science though, and he was such a fun and novel individual that capturing the spirit of who he was is perhaps best served by a series of personal anecdotes.
Field work with Ole and his family
My most intimate experiences with Ole were during field work at a remote camp on Lake Nerka, Alaska, in 1995, 1996, and 2000. For much of that time, it was just me and Ole and Ole's family, and it was one of the most memorable and rewarding of times for me. We worked long hours, ate good food, and had lots to drink, while playing games and watching nature. And we laughed and laughed and laughed. And nothing made him laugh so hard as watching his young kids teach me how to swear in Norwegian.
And then there was the electric drill. In 1995, Ole's main goal was to analyze the energy content of salmon and how it changed as they returned from the ocean to spawn in freshwater. To measure this energy content, we need to create a slury (or smoothie, if you will) of many many individual salmon. So Ole bought a Dewalt DW101 drill and hooked it up to a meat grinder. Powered by a generator, that amazing drill ran hours and hours each day making salmon smoothie. The result was a very influential paper (Hendry and Berg. 1999. CJZ 7:1663-1675). When he left Alaska, Ole proudly presented me with the drill, and it has been a valued household tool to this very day.
 |
| Ole's drill 27 years later. |
I feel compelled to comment again about how creative Ole was as a scientist. We had written a series of papers about how natural selection influences female reproductive life span in salmon. That is, a female couldn't just lay her eggs and die because she had to live somewhat longer to defend her nest site against other females who might dig on top and thus displace and destroy the earlier female's eggs. Yet no one had a good estimate of this selection on life span due to "nest superimposition." Ole had the amazing idea to inject females with colored food dye, which would bind to the eggs. Then we could dig up the females' nests to see how many of the colored eggs remained depending on whether another female later dug her nest in the same spot. This study was one of my favorites to this very day (Hendry et al. 2004. PRSB. 271:259-266).
 |
Ole digging up salmon nests (above) to look for dyed eggs (below). |
Visiting Ole and his family in Trondheim. I have many memories of my trips to Norway, but two come immediately to mind and put a big smile on my face. The first was when we took a long hike up into the mountains to camp at a lake Ole and colleagues were studying. We arrived at dusk and crowded into a small one-room cabin, with beverages all round. Not having seen an outhouse, I eventually worked up the courage to ask "Where do I go to the bathroom"? Ole laughed and shouted "Oh, just go anywhere over in Sweden." It turned out we were right beside the cairns that marked the border with Sweden, and anywhere past that border was the outhouse. It was my first visit to Sweden.
The other memory I want to share is how - when I spent several weeks staying with Ole and his family, they took great pleasure it introducing me to all of the traditional Norwegian "delicacies" - laughing at me as I tried them and telling stories about how Ole (and his kids) would weaponize food (mainly Surströmming) as practical jokes during military training (the whole barracks had to empty), at school (the police were called), and at athletic events (dropped on the opposing rowing team from a bridge above). Here is the menu and tasting notes that I generated from those days. Yes, they really cooked each of these dishes for me - mostly just to laugh at my reaction.
Sigurd Einum
In remembrance of my dear friend
and colleague Ole
It is now 30 years since I met Ole
for the first time, I was a bachelor student in biology, and he was faculty.
It’s funny how the teachers seemed old to me back then, he was only 38, and I’m
now 50! Anyways, I already then noticed how he was able to establish a rapport
with the students that few others did. I’m not sure how he did it, but I
believe his rather informal manner and a good (or bad?) sense of humor was part
of it. One of his more infamous pedagogic tricks was applied during lectures
when he noticed students closing their eyes and appearing to fall asleep. He
would then continue his lecturing in a normal voice while slowly approaching
the unsuspecting student, and once there yell something really load and
slamming his hands into the bench next to the sleeping head. Of course, he did
this with a smile on his face, raising laughs, and it usually ended well. As
another example, during a lecture on marine fish stocks in a class of about 100
students he, upon coming to a part about cod, got the students to participate
in singing a kid’s song that describes components of the life-cycle and ecology
of that species (Torskevise by Thorbjørn Egner). The thousands of students that
experienced this probably remembers a thing or two about cod, and at the same time
felt a bond to their professor that contributed to their motivation. While it
is tempting to try to replicate this, I’m afraid that only someone with Ole’s
personality can pull it off, and I would probably fail miserably.
 |
| Ole Kristian Berg in his element |
When I joined our department in
2007 I came to know Ole as a colleague. He was incredibly helpful whenever I
was having questions about how things were done in the department, and with
respect to access to labs and equipment. We quickly initiated collaborations
both with respect to teaching and science, and I have benefited greatly from
his broad knowledge about, ecology, life and basically everything. In 2010 he
invited me to join his expedition to Bear Island, which of course was
excellently organized, well stocked with equipment, food and drinks, and
required very little planning from my side. We also collaborated on our
freshwater ecology course, and the annual field course at remote Lake Snåsa was
always a highlight of the semester. I know Ole also appreciated these trips
with the students, and he continued to participate on the field course after he
retired. There we would teach coming generations of freshwater ecologists
procedures for fish and plankton sampling and processing, water chemistry, and
then at the end of a long day sit around the fire with a beer and watch the
northern lights together. These moments will be deeply missed.
A good friend and incredibly
generous and knowledgeable colleague has passed away, much too early. I had
looked forward to further collaborations, and not the least more Christmas
parties where he as usual was supposed to be responsible for the gløgg (mulled
wine), and being the last one to leave the party. That did not happen. Instead,
we will enjoy the good memories he has given us, and those are many! Ole
continues to live on in the hearts of collaborators and students. My deepest
condolences go to the family which has lost an amazing husband, father and
grandfather.
Thomas Quinn
 |
| Ole (left) and Tom (right) in Alaska |
Like
all others who knew Ole Kristian Berg, I was shocked and saddened to hear of
his passing, and extend my deepest sympathy to his family and friends. As sometimes
happens in our profession, we became acquainted when I got an unexpected
communication from him, indicating an interest in coming to meet and
collaborate with me. Thankfully, I encouraged him. One thing led to another and
30 years ago, in the summer of 1993, he visited Seattle, stayed with me, and
joined our field operations on Lake Aleknagik and Iliamna Lake, Alaska. The
attached photo is from that year. I was immediately taken with his personal
openness and friendly attitude, breadth of scientific ideas, and exceptional
work ethic. Then, in 1995 and 1996 he came again, with his wife, children, and field
technician, to work with Andrew Hendry, who was then my doctoral student. I was
fortunate that a house across the street, owned by our neighbors and friends,
Jim and Maxine Hinze, was vacant at the exact period when the Berg’s needed a
place to stay. Otherwise, I am sure I would have squeezed them into my house,
somehow! As always, he was lively, funny, kind, generous, and full of energy,
but also full of mischief and tricks. Ole kindly invited us to a barbeque but
then shocked us by revealing that he had smuggled minke whale meat into the US
and that was what we were eating! Watching them all repacking their great
volume of field gear among their bags to meet the weight requirements for
luggage at the Seattle airport was something I will never forget.
Ole’s
collaboration with Andrew Hendry resulted in a several important papers and a
great boost to Andrew’s career, but it also provided an endless series of
stories. The field camp where the Berg’s and Andrew were working, on Lake
Nerka, was a long distance from the main field camp on Lake Aleknagik, which
was accessible to stores for food, fuel, and other supplies. The Lake Nerka
camp had what seemed like more than adequate food but in short order the
unreliable old radio phone crackled with the news that they were running short
of essentials. How could this be? Well, it seems that they ate in a week the
supply of potatoes that would normally support a crew of that size for the
whole season! Over on Iliamna Lake, the largest lake in Alaska, I will never forget
wallowing in big waves in the middle of the night as we towed nets between two
boats to sample juvenile sockeye salmon and sticklebacks. We barely escaped
with our lives from the experience, but enjoyed a drink at 3 AM to celebrate
our brush with death on the lake.
I
raise a glass to a wonderful man – lively, loyal, experienced, wise, and kind.
Thrond Amundsen
Memories
of Ole Kristian
It was so
sad to hear that Ole Kristian had passed away so all too early. I’ve known Ole
as an always supportive and friendly colleague ever since I started my job at
NTNU, not so long after Ole was appointed himself, in the early 90s. I first
and foremost think of Ole as a kind man, always interested, always supportive,
always positive. These are qualities that don’t always thrive in modern
academia but they mean a lot, and Ole’s kindness meant a lot to me.
For many
years, up to Ole’s retirement and for longer than I can remember, I worked
closely with Ole on our first-year course in Ecology, Behaviour and Evolution
(BI1003). This is a special – and especially important – course for our
department: it’s the first course for new NTNU students of biology, including lots
of teachers and teaching assistants, and a large range of pedagogical tools and
activities. In short, a cool course but a logistic nightmare to manage,
requiring dedication ‘way beyond normal’. Ole put in enormous efforts and managed
the course successfully for several years until his retirement two years back,
to make it an enjoyable and successful course for students and teachers alike.
Being the two old-timers of the course, Ole and I had many interesting chats on
the smaller and bigger challenges of getting the course to work. I always felt
that Ole appreciated my input and that of others and did his very best to make
it a great course. Ole would have deserved much more praise for these efforts
than he got. Last year, the course and its teachers were awarded the Teaching
Excellence Award of the Faculty of Natural Sciences. Ole would truly have
deserved to be part of the award-winning team – we who took over built the
course on the foundation he laid down.
Ole was a
true ‘fish person’, with a huge knowledge of fish biology and fish management.
Other ‘fish people’ closer to his research can tell more about that then me.
For me, who started dabbling with fish from a bird background, he was a rich
source of knowledge and advice. And more importantly, he always showed a
genuine interest and appreciation – he always had a kind and supportive word.
I’m sure others can tell the same. To me, that meant more than he probably
realized himself.
My last
meeting with Ole was by accident, last fall, just outside the Natural Sciences
building. I had only occasionally seen him since his retirement. Ole was in a
good mood as always, and curious to hear about my stuff, including how things
had worked out with my own group’s field work last summer (fortunately, for
once, quite well!). On my part, I was curious how things were for him, and what
he was up to at campus. Turned out he was on his way to talk to BI1003 students
about how to present the projects they were about to complete, contributing as
a ‘volunteer’. That’s typical of him – he loved that course, he loved engaging
with students, and he was a very unselfish man, happy to help out when asked.
On the private side, he told that he and his wife had spent the whole summer
traveling Iceland, enjoying its splendid nature – and I believe culture, too.
It sounded like they had had a great time up there, and we shared memories of
Iceland’s natural riches.
When Ole
has now so sadly passed away, that last meeting stands fresh in my memory, as
typical of Ole – friendly, supportive, with a true love for nature and for
educating the next generation about the value of nature. It makes me think that
Ole lived his life to the full as a biologist, teacher, nature lover and family
man until the very end. Even if that end came all too early.
 |
| Ole (left) was always happy - but never more so than when up to his wrists in fish guts. |
Gunnbjørn Bremset, Jan Grimsrud Davidsen, Tor G. Heggberget, Sten Karlsson, Line Elisabeth Sundt-Hansen, and Eva Marita Ulvan
It is with great sadness we
learned of Ole Kristian Berg's untimely passing. Most of us knew Ole Kristian
from his time at the Zoological Institute at Rosenborg campus, where he devoted
much of his research on the landlocked salmon (småblank) in River Namsen and
River Mellingselva. It is especially our research collaboration on the småblank
population in Upper Namsen that brought us together, where we mapped the status
and distribution of småblank, as well as its unique genetical and habitat
use. Småblank was a natural study subject since Ole Kristian's father, Magnus
Berg, was the first to describe the special salmon variant only found in the upper parts of
the Namsen watercourse. Ole Kristian was very keen on preserving the original
name of the landlocked
salmon in Namsen, and it
was therefore a great academic victory
when "småblank" finally became the official nomenclature a few years
back. Although småblank had a special place in his heart, there were many other
areas of research that Ole Kristian worked on. Already in the 1980s, he focused
on juvenile salmon that resided
in atypical habitats, and together with graduate students, investigations were
made in lakes on the west coast and in Namdalen, before later focusing on
deeper lotic habitats in rivers in Nordmøre and Trøndelag. Ole Kristian was a
pioneer in using modern analytical methods and was the first in Norway to
conduct energetic analyses of juvenile salmonids in rivers such as River Homla
and River Stjørdalselva. Many Norwegian fish researchers became interested in
fish biology after Ole Kristian's lectures, and the number of students who have
had him as a supervisor has gradually become extensive. In terms of personal
qualities, we will especially highlight his good humor, infectious laughter,
and unyielding optimism, and that he was very caring and generous towards his
students and colleagues. His many antics during festive events at Rosenborg and
the Natural Science Building are still being talked about, and we will never
forget his performances with specially made dentures and glasses with special
lenses. Ole Kristian will be deeply missed as a professional, colleague, and
friend - and we will forever keep the memory of him in our hearts.
Ole's Most Cited Papers